
In almost every manufacturing operation, regardless of the materials used, the surface finish is usually the last step, and possibly the most important one in creating a solid and durable product. In our everyday lives, we’re always in contact with several appliances, equipment, and products that either have a finished surface or require parts with a surface finish. This is why such processes constitute a necessity in most industries.
The several surface finishing processes all share a similar functionality of enhancing the surface of a substrate. As such, the material’s surface is rendered more uniform, with a smoother texture. However, these different methods each have specific characteristics and different uses, depending on how they alter the surface of a product in terms of corrosion and wear resistance, texture, coloring, and coating. Besides the technical advantages, a consistent surface finish can also add value to a product. It enhances the aesthetic appeal and overall appearance. As for components and industrial or mechanical parts, it improves performance and durability as well as the parts’ fit for usages requiring tight tolerances.
Most of the standard finishing processes in industrial usages are usually applied to a wide range of soft and heavy metals like cast iron, steel, and aluminum. With the recent developments in manufacture and technology, industrial and machining surface finishing techniques are now also applicable to more brittle materials that have previously been finished manually. These materials can include pitch, ceramic, glass, some types of stone as well as plastic.
As diverse as surface finishes are, they can essentially be defined as a process employed to alter the surface of an item (or substrate) to achieve a certain property. The exact desired surface finish is determined by specific parameters. Roughness is quantified by the deviation between peaks and valleys in the surface. Waviness, which is rather similar to roughness, is strictly defined as irregularities wider than the average roughness sampling length. The final parameter is lay, and it refers to the directionality of the surface’s dominant pattern.
Regardless of the preferred method or process, surface finishing, in general, requires the utmost accuracy and in some cases constant monitoring of some parameters. As such, this is a very delicate process. By now, there are several standards and criteria regulating most surface finishing processes.
Table of Contents
Charts And Symbols
In CNC machining, there is a wide range of surface finishing charts that can be employed. The most widely used one is the roughness chart, which shows a graphic representation of the surface’s roughness. This can be used to help determine the best machining process for a given surface. Along with a standard chart for different surface finishes, which shows the measurements of different surface textures, both can be used to determine whether a surface is within the desired tolerances. As a best practice, using this chart along with the roughness chart can help operators understand exactly what parameters they need to stay within.
These charts often include several guiding symbols, which can be present in technical drawings as well. These symbols indicate the appearance of a surface after it has been machined. Each of these symbols has a specific meaning and can be used to describe different aspects of a surface finish:
- Ra – Average Roughness: Ra is a measure of the average roughness of a surface. It is a unitless quantity that expresses the average height of the surface irregularities, or peaks and valleys, over a given length.
- Rz – Average Maximum Height of the Profile: Rz calculates the average difference between the five largest peaks and valleys. This measurement is done using five different sampling lengths, to eliminate any errors. Ra is quite insensitive to some extremes, so this calculation is very helpful.
- Rmax – Vertical Distance from Peak to Valley: Rmax is more sensitive to anomalies such as burrs and scratches that cannot be seen only by Ra.
- RMS- Roughness average magnitude surface: The RMS- roughness average magnitude surface is a surface that is used to measure the average roughness of a surface. This surface is created by taking the absolute value of the surface’s RMS roughness magnitude.
- PE- Profile roughness: The profile roughness of a material is a measure of the size and distribution of the surface irregularities. It is usually expressed in terms of the height of the largest peak or the depth of the deepest valley on the surface.
- PS- Profile smoothness: Profile smoothness is the average area of flat surfaces in a profile of a surface. It is usually expressed in terms of the breadth of the flattest section of the surface or the thickness of the thinnest portion of the surface.
- PT- Profile tolerances: Profile tolerances are the allowable deviations from a nominal profile. They are important to ensure that parts fit together correctly and perform as intended. Profile tolerances are specified in terms of the maximum allowable deviation from the nominal profile at any point along the part’s length.
Learn more about Surface Finishes Chart for CNC Machining
Symbols and Standards
Surface finishing symbols on the other hand serve a slightly different function in industrial operations. They visually designate the results of a machining process to specify the processes employed for a specific surface finish. The main purpose is to help find and identify pieces that have been machined by specific processes without having to research individual machines. This can save a great deal of time and effort for both customers and suppliers.
These symbols can also indicate the quality of a surface. They can be used to describe the appearance of a surface or the feel of a surface. Different parts of the world have different surface finish symbols and standards. The ANSI standard is commonly used in the United States, while the ISO standard is more popular elsewhere in the world.
The ISO surface finish symbols
The ISO surface finish symbols provide a standard for the surface finish of manufactured components. Each symbol is used to communicate the surface finish to engineers and manufacturers and can be used as a reference for the surface finish of different items. There are a variety of different symbols, each representing a different surface finish.
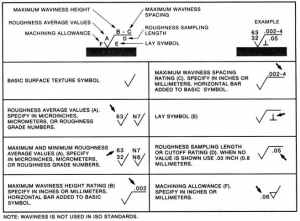
ANSI Surface Finish Symbols:
The American National Standards Institute (ANSI) has a set of surface finish symbols used to indicate the quality of a surface. Most manufacturers and buyers are familiar with these symbols as standard for surface finish quality.
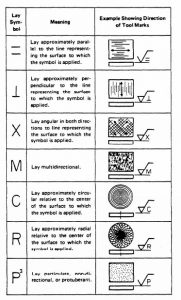
Learn more about the Different Types of Surface Finishing Symbols and their Meanings
Mill Finish
Surface finish processes tend to be the final stage in most manufacturing operations. Before the application of any finishing method, the material comes into the chain in its raw form. Metals are by far the most common materials for most industrial applications. The raw material is prepared for the surface finish after exiting the milling process. As a standard in many industries, the raw grainy texture of the raw, industrial-use ready metal is usually referred to as a mill finish.
In most manufacturing operations, the last process a substrate goes through before the surface finish is milling. The overall shape of an industrial part is created in this stage, leaving a rough and coarse texture to the substrate. This is most common for aluminum and most metals but is also a standard process for other materials used in subtractive manufacturing.
This rough and unpolished texture lacks brightness and has low reflectivity. It can also contain some trace amounts of mill oil or oxidation spots on the outer layer. Mill finishes are usually considered raw or unfinished, requiring a surface processing step to create the final appearance of the component. Essentially, a mill finish is almost a requirement for surface finishing processes, providing the accurate measurements, and precise angles of a component before applying a finish.

Importance of a Mill Finish:
Although a mill finish is an unfinished component texture, it is very sought after by manufacturers and builders. Besides being the standard form in which raw metals enter the manufacturing process in several industries, it also offers a greater degree of customization for manufacturers and designers. In most industrial uses metal parts or bars are processed into a coherent component, or final product comprising different pieces. Likewise, the optimal surface finish is determined by the final usage of the product, among other factors, which significantly guide the sealing and coating of the material. This arrangement method allows for a consistent appearance and functionality of the final product or component.
Characteristics of a Mill Finish:
Although a mill finish is a recognized standard, there are usually slight differences between the different metals as well material production processes. The most common mill finish metal is aluminum. At this state, mill finish aluminum will usually have a very thin oxide film, which is common for both extruded and rolled aluminum. Oxidation, in general, can be present in most mill-finished metals, which will later be removed through a chemical process. This is the case also for stainless steel, which usually exits the mill with very low luster, sometimes even a dark coloring on the surface. The metal that comes with the thinnest oxidation coating after rolling is by far sheet metal.
The resulting mill finish of bars of raw metal depends on a variety of factors. Besides the subtle differences between the mill finish of metal bars produced by several mills, there are slight differences between each stock produced in the same batch. The process itself, milling or extrusion, will greatly affect the characteristics of the mill finish. For example, in rolling mills, hot-worked metals will usually exhibit an oxidized surface, with a dark, grainy, and non-reflective finish. This is different from what extrusion mills produce; metal bars with die marks along the stock’s length. Recent developments in manufacturing have enabled mills to enhance the surface finish of their raw metals, by adding polishing rollers that can add brightness and a finer finish to the metal’s surface.
Surface Finish Processes:
Several factors determine the specific choice of a surface finish method or combination. The most prominent of all is the desired surface finish. However, other conditions like tight tolerances, the substrate’s material as well the final usage of the component can often determine this choice of surface. Surface finish methods in CNC machining and subtractive manufacturing usually involve some mechanical methods; pressure and surface friction usually constitute the base of surface finishing. Chemical methods are also quite common in surface finish processes. Besides the purely chemical methods, even mechanical methods often include some mechanical manipulations.
Learn more about Mill Finish
Grinding and Lapping

Two of the most widely used machining techniques in surface finishes. This process usually applies to different materials, but most commonly hard ones like metal and glass. Although there is a difference between grinding and lapping, the two terms are used interchangeably, as both processes are quite similar and are often complementary. The two methods both involve friction, at normally low speed, to remove material from the high points of a workpiece and smoothen the surface.
Machining lapping is optimal for products requiring precise flatness, parallelism, with measured requirements in terms of consistency and finish. This method is characterized by precision in terms of surface roughness levels. It’s an age-old method that technology has come to render more precise, and relied upon by many industries for several usages.
Grinding and lapping can also be used to remove surface defects through friction, usually with the help of a flat lapping machine. The surface of the workpiece rubs against the lap of the machine (usually a surface of iron), through a layer of a mixture composed of abrasive material like emery, silicon carbide, or diamond. In most CNC usages, lapping machines are high-precision units and produce a very fine and consistent surface, free of even microscopic imperfections. Although machining lapping usually produces a uniformly smooth and flat surface, it can also produce concave and convex surfaces.
Difference Between Grinding and Lapping:
There are several similarities between the two processes and are both employed for the same effect, but they differ in some key aspects. Machining lapping is a loose abrasive process (LAP) and relies mainly on the abrasive mixture. This liquid cutting tool, or a Slurry, is a mixture of abrasive grains and a water-based or oil-based solution. In a batch process, the abrasive mixture is spread across the flat lap plate. The workpiece is held in place using the retaining rings. The parts are then dragged across the lapping plate surface in a circular motion.
Grinding differs in using an abrasive wheel (or grinding wheel) usually composed of the same abrasive material used to make the Slurry mixture, and conform to the same parameters of abrasive grain size and friability. The two processes can also differ in terms of material. Grinding is mostly compatible with harder materials like metal and glass. However, machining lapping is more compatible with brittle materials like pitch, ceramic, and some types of stone.
Throughout the grinding or lapping process, surface accuracy (or surface flatness) and roughness are closely monitored. Roughness is measured usually in microns, or by quantifying the averaging the variances of peaks and valleys (Ra value). while accuracy, or flatness, is measured in Helium Light Bands (HLB). Light interference is now an increasingly precise measuring technique, employing lights with the smallest wavelength.
The required levels of surface roughness determine the choice of the abrasive. Abrasive particles of smaller grain size (less than .025 micron) like aluminum oxide are more conducive to very fine, almost undetectable micro scratches, and produce a fine finish.
Brushing
The brushing process is usually most commonly used for metal surfaces, in industrial applications. The process begins with cleaning the substrate or workpiece. The surface is then sanded with fine-grit sandpaper to remove any scratches or blemishes. Next, a metal brush is used to apply the desired finish to the metal surface, moving in a circular motion at various speeds, depending on the desired finish and material. Finally, the surface is rinsed to remove any metal residue.
The metal brush can be used to apply a variety of finishes such as polished metal, chrome, and nickel plating, brushed aluminum finishing, steel brushing, and more. The type of metal being polished by brushing, and the desired surface finish both determine the thickness of the finish applied. The metal brushing process is also known as a mechanical polishing process.
Process Brushes:
There are many types of metal brushes available on the market. For instance, most metal finishing tools have either a round or rectangular shape, with varying sizes. Additionally, some metal finishing tools include long and thin synthetic bristles, usually applied to apply high surface contact. These synthetic bristles are an optimal choice for most metal polishing applications as they can produce even fine surface textures.
Abrasive brushes are widely used to finish surfaces. The abrasive tools used for brushing can create perfect finishes on uneven surfaces, resulting in a beautiful brushed metal finish. The several surface finishes achievable by brushing are quite diverse and can be adapted for a broad range of applications. Abrasive brushes are high performance. Their main advantage is the ability to produce exceptionally smooth finishes on even the most irregular surfaces.
Applications:
Brushing produces a smooth finish with a recognizable texture. Besides its wide range of applications, brushing can also be applied to preserve metal and protect surfaces from scratches and damage. The technique is typically used in a variety of applications, among which are:
- Deburring: is the removal of sharp edges and burrs from a metal surface. This is achieved with metal finishing brushes used specifically for CNC deburring. They can also help remove sharp edges, and prevent rust and corrosion of metal parts.
- Edge blending: Another application of brushing finish is to smooth out the transition between two different surfaces. This can often make the edge more uniform and prevent it from causing a tear in the material with which it comes in contact. Using the brushing finish technique works best for creating sharp, crisp edges.
- Cleaning: Abrasive brushes are effective in surface preparation for other surface finish processes, or a more exhaustive brush for effect. These brushes can remove dirt, film, rust, weld slag, and other contaminants from a surface.
- Roughing: The brushing finish can also be applied to create rough surfaces. The roughening leaves less surface area to catch dirt and debris and is easier to clean.
Learn more about the Brushing Finish Process
Polishing

Metals are some of the most resistant and reliable materials available. They’re versatile in their industrial and mechanical applications due to their strength. This makes them often in contact with degrading elements or conditions, like air, heat, and high electrical current density. This need to preserve strong materials from corrosion and wear is what makes polishing one of the most widely applied surface finish processes.
This process can contribute significantly to the aesthetic appeal, functionality, and is optimal for components with tight tolerances. Polishing also prevents contamination of metal parts and removes oxidation. Surface polishing has been in application for many centuries. It has also been used on a variety of materials, ranging from copper and metals to glass and wood. Industrial applications of this process differ in their focus on more solid materials, notably metals.
The process is mostly mechanical in the sense that relies on friction to make the substrate’s surface more uniformly flat, smooth and consistent. It applies the same principle as grinding or lapping but is still different in many regards. Depending on the specific metal used, polishing finishes come in several varieties, and to differing effects. Polishing can often keep a surface in excellent conditions, yet requires specific measurements and design guidelines to harness its full potential.
Polishing and Buffing:
The surface polishing process is sometimes also referred to as buffing, although there is a slight difference between the two. Buffing is usually the final or latter stages of the process. This is designed to improve the small imperfections in the surface, not visible to the naked eye. The general process, or principle, of polishing, has now expanded and can be adapted by using newer methods like electrolytic polishing, or chemical polishing.
Polishing usually begins with the usage of a rough abrasive of higher grain size. This depends on the material of the workpiece and its surface. The aim here is to remove the major imperfections; convexities, scratches, lines, or pits. The process begins with the application of an abrasive, in an oil-based mixture, polishing pads and buffing wheels, and later on, employs abrasive grains of a smaller size. This transition from larger grains to smaller ones refines the minute valleys and peaks, and manufacturers often use this method to obtain a reflective surface, or mirror finish, using high-speed polishing machines.
The currently used polishing methods can apply to more materials. The most used materials in many subtractive manufacture and CNC machining usages, although more brittle metals like brass, silver, and gold are also polished, as well as plastic. The substrate’s material and the desired finish largely determine the choice of buffing compound (abrasive), the process is organized into three main stages. Rough, Initial buff, and final buff, each using finer and finer abrasive grain size, significantly impacting the polish finish.
The wafers, or buffing wheels, differ in terms of material as well as usage. Each type is used for specific steps of the process. Canton flannel, string, and sisal fiber are amongst the most commonly used. Similarly, the buffing compound is also specified for distinct usages and is categorized by color.
Precision Requirements:
As a detail-reliant task, polishing and buffing rely on several decisions and choices that are determined by the required finish. The choice of abrasive type and buffing wheel are prime examples. The surface finish is vulnerable to many subtleties of polishing. There are a variety of buffing wheels, made from different abrasive materials, but abrasive discs with polyester fillings are preferred as they can achieve a more uniform and consistent finish.
Each abrasive compound is specific to a distinct material of the buffing wheel. However, it is possible to use a buffing wheel for different materials as long as it is thoroughly cleaned from the compound residue before usage. Like most surface finishing processes, polishing too requires pre-treatment. The surface should be cleaned, and have any lacquer removed to ensure better contact between the substrate and the buffing wheel.
Learn more about Polishing
Bead Blasting

The bead blasting process consists of propelling beads towards a workpiece or substrate through small-gauge holes in a cup. This essentially removes the outer material away from the substrate and takes surface coating off metal objects. There could be several expensive tools and appliances requiring the removal of a surface coating or oxidation layer. This process smoothens surfaces to ensure a better contact with other materials.
Bead blasting usage is not restricted to metals. Manufacturers may also use it on other materials such as plastic, glass, and rubber to leave a flawless surface finish. The beads are composed of abrasive materials, propelled to clean or etch a surface. The beads are accelerated by a gas such as air, nitrogen, or carbon dioxide. They hit the surface at high speeds, dislodging any dirt or debris. The process can also be used to create a textured surface. The beads can also be made either of glass, plastic, or metal.
Applications and Materials:
Bead blasting can be used on a variety of metals. The metal is placed in a chamber and small beads are fired at it at high speeds. High-speed abrasion plays an important role in this process and constitutes the basic principle behind this method. A blasted surface can usually exhibit a matte finish, after the removal of rust. This method is usually done in preparation for painting or powder of aluminum, titanium, and other metals.
Bead blasting aluminum is a popular technique for cleaning and polishing aluminum surfaces. Several factors make this technique preferable for most manufacturing operations. Bead blasted aluminum is optimal for a wide variety of applications. It is strong, durable, and lightweight, making it perfect for use in manufacturing, construction, and other industries. Additionally, bead-blasted aluminum is resistant to corrosion and wear and is optimal for outdoor applications. The correct grade of aluminum is largely determined by the required surface finish. Phosphated finishes and anodized ones are the most common
Advantages and Disadvantages:
The process of bead blasting has various advantages. However, it can be very abrasive and can damage the surface if not done correctly. It can produce a matte finish and is quick and cost-effective. Coupled with the durability and long-lasting effect of bead-blasted surfaces, which are effective for high traffic areas and outdoor usages, bead blasting is optimal in many ways.
This is however offset by the risk of overspray and uneven surface, often caused by multiple passes. This method requires highly skilled and trained workers to produce efficiently and accurately and is usually followed by a final buffing.
Learn More About What Is Bead Blasting and How Does It Work?
Anodizing

Anodizing is a metal finishing process, which creates a thin, hard, and corrosion-resistant layer on the surface of the metal. Aluminum is the most commonly used material in this process. The lightweight of the material and the corrosion resistance of an anodized finish make this combination perfect for aircraft construction. Although this process has mostly been applied to aluminum parts, it can now also work for a variety of materials such as zinc, titanium, magnesium, and zirconium. Because of the electrolytic nature of this technique, it does not work on ferrous metals, or metal bars containing significant quantities of iron, as it exfoliates when anodized. Iron forms a layer of oxides or rust.
The main advantage of anodizing is adding an anodic oxide finish, which works as anti-corrosion protective later. To create an anodized coating, aluminum is first immersed in an acidic electrolyte solution that reacts with the surface of the metal. The electrically charged anodizing solution provides perfectly monitored conditions for maximum accuracy. The substrate is then subjected to an electrochemical reaction which causes an oxide layer to form on the surface.
The regular anodizing process may not always provide as reliable as surface finish and protective film, notably the case for some types of aluminum alloys. For this, a specialized alumite treatment liquid is used, in a method known as precision anodizing.
Process Details:
Like most surface finish processes, the first step is surface preparation. Cleaning the surface from impurities is standard in many manufacturing operations and CNC usages, as it can greatly hinder the surface finish if done improperly. A solution is usually applied to the material for this process, referred to as a Desmut solution. The latter has mostly been composed of Nitric acid, but this component is no longer applied due to environmental concerns.
After the cleaning process, the material is placed into a bath of an electrolytic solution. When a direct electrical current is applied, it creates a positive electric charge in the material (usually aluminum) and a negative charge in the solution’s electrolyte plates. The anode for this operation is the material, or substrate, while the cathode is the aluminum plate of the electrolytic cell. The oxygen resulting from this reaction enables the substrate to bond with oxygen ions, creating aluminum oxide.
The acidic solution in which the substrate is dumped slowly dissolves the aluminum oxide, forming at the surface. This balance forms nano-pores on the surface. The solution reaches the substrate more effectively and increases the thickness of the protective layer. This would facilitate the absorption of the dye later on. The anodizing process is usually followed by a sealing stage to keep the dye layer intact.
Recyclability of Anodized Metals:
Anodizing has long been associated with aluminum, although it can apply to other non-ferrous metals as well. Anodized aluminum is largely considered environmentally-friendly. This is true of anodized metals in general, as they are easily recycled, which saves the energy required to mine and produce more non-ferrous metals. This quality is also accentuated with reinforcing properties of the anodizing process, which makes anodized metal parts durable and reusable. Compared with other metal surface finish methods, anodizing is both cost-effective and can significantly enhance the life cycle of aluminum parts. Anodizing essentially accentuates the environmental benefits of aluminum.
As a water-based process, anodizing does not use any VOCs (Volatile Organic Compounds), toxic organics, and no solvents. The colors generated through anodizing use an insignificant amount of dye. In addition, anodizing produces no hazardous waste.
The compatibility of more and more non-ferrous metals with the anodizing process, will increase the number of recyclable metals used in industry and manufacture, and decrease the industrial demand for the durable metal. The environmental benefits of anodizing exceed the impact it has on a component’s life cycle, but constitute a reliable and environmentally safe industrial process.
Electroplating

Electroplating is a method of plating metals onto other materials using electricity. It is commonly used to create decorative finishes on metal objects such as jewelry or automotive parts. The general process consists of immersing the object in a bath of an electrolytic solution, at high voltage using two electrodes; one at each end of the substrate. Ions from the electrolyte are then deposited onto the substrate’s surface, creating a thin and electrically conductive layer. The object can then be removed from the bath and rinsed with deionized water to remove any remaining electrolyte.
Electroplating has become very important in modern manufacturing processes because it provides several advantages over conventional methods. As an efficient and cost-effective method, compared with conventional methods, it allows for a higher quality finish with fewer imperfections on the surface.
Process Details:
The electroplating process is an important part of manufacturing metals and other materials that are typically found in electronic appliances. The process relies on an electrical current applied to the metal and conducting it to the substrate. The first step of the process is immersing the workpiece in an electrolytic cell; a bath composed of a mixture of chemicals, metals, and other substances.
Afterward, the electrodes are submerged in the electrolyte solution, and then connected to a power supply, and through the target substrate. The third step in electroplating is a cathode protection stage, which prevents corrosion during plating. Finally, the fourth step is called electrolysis, where an electric current causes ions to be released into the electrolyte solution.
Different Materials: Electroplating can be applied to various metals besides aluminum. Steel, for instance, is a widely used metal. Largely because of its strength and durability, it is used for outdoor application, but it can also get weak if exposed to a corrosive environment. Many industries rely on steel to ensure the quality of their products. The properties of steel however can be manipulated per the strength of its coating. As such, steel with aluminum plating can remain strong for years.
Materials:
Copper is a versatile and durable metal, which has been in use for thousands of years. It is generally applied as a conductor of electricity, and heat, used in the minting of coins, and production of jewelry and utensils. Copper is also corrosion-resistant and suitable for usages requiring solid and durable material. When copper is being soldered, the base metal layer (copper in this case) often has a second layer applied to it.
Aluminum electroplating has been a commonly used technique in the production of glassware for some years. This process gives the glass a strong protective layer and is an interesting way of making the surface of glass-made parts electrically conductive.
Learn More About Electroplating
Alodine

Alodine is a trading name for a chemical compound that is used as a corrosion inhibitor. It is a water-soluble, organic compound used to coat metal surfaces to protect them from corrosion. Alodine is mostly distributed as a powder, which can be mixed with water. There is also a pre-mixed variety available. This process is safe to apply on all types of metal, including stainless steel, copper, aluminum, brass, and bronze.
Alodine is used primarily as a corrosion inhibitor. It is more of a protective coating in this sense. The process is more akin to chromate conversion coating; a common method used for metals. Alodine generally forms a protective layer on top of the surface and can harden to the same consistency as the base material. Alodine not only prevents corrosion but also makes a product more durable.
Although Alodine chemically embeds into the surface of the workpiece, creating a corrosion-resistant coating, it does not alter the existing characteristics of the metal. An Alodine finish provides an improvement in corrosion resistance over the original metal, and can even render the material resistant to hazardous elements like chlorine gas.
Alodine and Different Metals:
A clear alodine coating is applied to aluminum parts to protect as a corrosion protection measure (anodic oxidation). The alodine aluminum chromate coating is formed by immersing the aluminum component in a bath of chromic acid and then rinsing it with water. For this application, the chemical solution consists usually of chromic acid. Clear alodine forms a layer when sprayed, and hardens when dried. It is often used for aluminum window frames, wheels, and grills.
Stainless steel parts are coated with a clear alodine coating that is similar to the coating used on aluminum. This coating creates a uniform, nonporous surface. The chemical reaction occurring between the steel surface and the alodine, at a temperature of 600 degrees Celsius is the main catalyst of this transformation. The coating is then removed after it forms and is cleaned. This process forms a protective barrier on the surface of the steel.
Benefits and Limitations:
Alodine finish is optimal for metal parts, and boasts numerous benefits. It offers optimal corrosion and wear resistance, with a protective coating that is compatible with several types of metal; steel, aluminum, stainless steel, and titanium. The surface finish and protection of alodine are also durable and can withstand harsh environments and conditions. As aluminum and heavy metals are mostly used in industrial operations and high traffic usages (aircraft, construction, industrial equipment), alodine finishes are quite vital to several sectors. The alodine finish is also cost-effective and relatively safe; it is used in the medical industry and doesn’t rely on any polluting or toxic materials. Alodine finishes have excellent adhesion to metals and are recycled easily.
These advantages are however offset by the limitations of this method which include the alodine solution, which can often be toxic to the skin. As far as industrial usage goes, alodine finishes are protective but less durable than other methods. The coloring that this method offers can often fade with time, especially for components used in high-traffic areas or outdoors. The choice of an alodine, compared to other methods, relies solely on the intended use of the final product.
Learn More About Alodine vs Anodize
Powder Coating

During the last few decades, powder coating has become somewhat of an industry standard. Especially for materials like metal, powder coating is now the optimal surface finish process for a wide variety of items, ranging from home appliances to industrial and automotive parts. Although powder coating has mostly been used for metal, it can also cover other materials, notably plastic, carbon fiber, and MDF (medium-density fiberboard).
In powder coating, a fine layer composed of free-flowing powder is applied to the surface of the substrate. The main process consists of propelling the powder particles using an electrostatic charge. The powder then embeds into the microscopic pores and cracks in the surface. The powder is then fused with heat and pressure, forming an even coat on top of the base material. The powder is a generally preferred method of surface finish and dye as it’s much cleaner and produces no volatile organic compounds (VOCs). This method significantly decreases the cost of pollution control. Another advantage is that powder coating produces a much thicker and firmer coat, compared to paint.
General Process:
As in several processes, the first is the preparation of the surface. The substrate’s surface is cleaned, and its impurities are removed through different methods. Depending on the substrate’s material, different methods can be applied. Heat-sensitive materials like plastic have only recently become compatible with this method, largely enabled by the breakthroughs in plasma pretreatment.
The application stage consists of the actual coating. This process usually requires an electrostatic spray gun. As the powder is sprayed towards the substrate, the electrostatic charge accelerates it towards the workpiece.
The final step is the curing or heating stage. The powder is exposed to heat, dissolving it into a uniform layer. After cooling, the coating solidifies and forms a consistent protective layer. Some solid materials like iron are usually preheated before coating, which helps avoid some coating inconsistencies later in the process. Curing ovens are usually required for this process; a specialized industrial oven capable of reaching very high temperatures ranging between 162 and 232 degrees Celsius (325 to 450 Fahrenheit). For this range, most materials require a curing time of between 10 minutes and an hour.
Metallic and Protective Coatings:
Metallic coatings can also constitute a distinct method, as they serve mainly a protective purpose. A metallic coating essentially transforms some of the properties of the substrate. The use of metallic materials, usually zinc alloy or aluminum, creates a very solid coating on the surface. There are several common methods to this process. Usually, metal or iron are coated with zinc, by passing the material through molten zinc at a temperature of 460 C (860 F). This is known as hot-dip galvanizing.
- Thermal spraying is also a very common powder coating method. This process relies on coating the surface with a finely divided, semi-molten metallic material.
- Sheradizing is another form of metallic coating. This method consists of heating the substrate in an airtight container with zinc powder. As a thermal diffusion process occurs, the zinc-iron alloy forms a protective layer, embedded into the substrate’s surface.
Common Applications:
Powder coating is a very common method for most manufacturing operations in different industries. The strength of the outer coating it provides, and resistance to corrosion, coupled with cost-effectiveness and ease make it one of the most reliable surface finish processes.
Several automotive components rely on powder coating. This process applies to anything from door handles and bumpers to brake components and battery trays, as well as medical equipment. This is largely due to the anti-corrosion properties of this method. Powder coating applies to some electrical components as well, notably connectors, lighting fixtures, and electrical junction boxes. Nowadays, many types of equipment, machine, and appliances go through this process, it has become the standard in other industries.
Some forms of metallic coatings serve only as a protective layer for the substrate, countering corrosion, chemical wear, and withstanding harsh environments. This is most commonly applied to steel.
Characteristics Of Surface Finishes
Corrosion Resistance:
This is a common trait shared by all of the above-mentioned surface finishing methods. Yet, some techniques can deliver this trait more than others, depending on the substrate’s material. Metal components are usually the most demanding in terms of a protective surface layer. For most applications, metal components are usually finished in a method that provides a protective layer. Polishing and anodizing are the most commonly used for this quality. Generally, electrolytic or chemical-based finishing processes offer better surface protection and corrosion and wear resistance. Mechanical finish methods are mostly used to prepare a solid surface compatible with the requirements of a chemical or electrolytic process. The powder is also a commonly used method for this purpose. This method is however mostly compatible with usages requiring specific coloring effects and textures, usually unattainable with other methods. The additional layer produced by powder coating has protective qualities, providing an impermeable coating, which preserves the material from decay.
Surface Smoothness and Tight Tolerances:
The technology and industry standards available today enable electrolytic and chemical surface finish processes to be more precise and highly customizable. Yet, in terms of surface accuracy and precise roughness levels, mechanical methods are still the optimal choice. Processes such as machining lapping, polishing, and brushing can only remove a limited amount of material from the workpiece. This is a common trait of detail and accuracy-oriented methods. Brushing, for example, can be used on complex surface shapes, but the vast array of brushes and customizability in terms of speed and direction allows for it to obtain specific surface textures that would have been very complex to produce with another technique.
Aesthetic Appeal and Colors:
In most industries, the surface finish provided by anodized aluminum is instantly recognizable. The smooth texture and embedded coloring are defining characteristics of this method. This is because anodizing can create coloring effects as part of the process, without requiring an additional coating of paint or dye. Powder Coating however is more distinctive compared to other methods. It is the industry standard for customized coloring. It produces a thick and firm coating, embedded into the surface. Most manufacturers rely on a combination of methods to produce a final surface finish. Mechanical methods are used to obtain the finest and most accurate surface, which will then be coated.
Conclusion
Surface Finish Methods are numerous and different in their applications, characteristics, and needed equipment. Yet, it is still one of the most demanded services. Almost every appliance, tool, or machine requires a surface finish or contains parts requiring tight tolerance and specific surface textures. As such, the knowledge and technology enabling this process is constantly developing, creating new ways, and transforming the ones used today. Surface finishes in general are still a necessity. Whether a certain product requires tight tolerances or specific surface textures, surface finish processes in most subtractive manufacturing applications are designed to enhance specific qualities in the treated material, making it greatly compatible with any type of usage.
